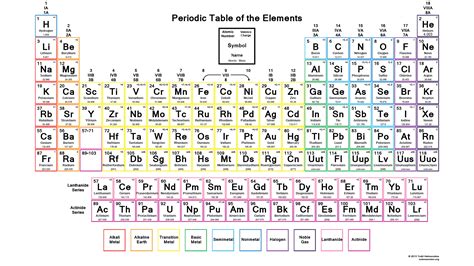
Understanding periodic table charges is essential for grasping the fundamental principles of element reactivity and predicting chemical behavior. The periodic table, organized by atomic number and element category, not only identifies elements but also encodes crucial information about their chemical properties, including oxidation states, which are pivotal for chemical reactions.
Practical applications of periodic table charges extend from simple compound formation to complex industrial processes and biological functions. A deep dive into these charges reveals the underlying mechanisms driving chemical interactions and reactivity.
Key Insights
- Understanding periodic table charges helps predict element reactivity.
- Valence electrons, dictated by periodic table position, influence oxidation states and reactivity.
- Knowing these charges allows for effective manipulation in various chemical and industrial applications.
Valence Electrons: The Backbone of Chemical Reactivity
Valence electrons, the outermost electrons in an atom, are the primary determinants of element reactivity. The periodic table’s layout reveals the number of valence electrons for each element by its group and period. For instance, elements in Group 1, such as lithium (Li) and sodium (Na), have a single valence electron, leading to high reactivity as they readily lose this electron to achieve a stable electronic configuration.Oxidation States: Predict and Control Reactivity
Oxidation states, or oxidation numbers, are a direct reflection of periodic table charges. They represent the charge an atom would have if all its bonds were ionic. Elements in the same group have similar oxidation states, but these can vary based on the element’s specific electronic configuration. For example, sulfur (S) in Group 16 can exhibit oxidation states of -2, +4, and +6, showcasing its versatile reactivity. This variability in oxidation states allows sulfur to participate in diverse chemical reactions, such as forming compounds like hydrogen sulfide (H₂S) or sulfuric acid (H₂SO₄).What role do periodic table charges play in biological systems?
Periodic table charges are fundamental in understanding biological processes, such as enzyme-substrate interactions and the behavior of ions in the human body. For instance, calcium (Ca) plays a critical role in nerve function and muscle contraction due to its specific charge and ability to form stable complexes with phosphates.
How do periodic table charges influence industrial processes?
In industrial chemistry, understanding periodic table charges is essential for designing efficient processes like catalysis and material synthesis. For example, platinum (Pt) groups' high oxidation states are leveraged in catalytic converters to reduce harmful emissions by facilitating chemical reactions.
This detailed understanding of periodic table charges enables chemists and engineers to predict and control the reactivity of elements, thereby advancing both scientific discovery and industrial innovation. Mastery of these principles is crucial for anyone involved in chemistry, whether for academic purposes or professional applications.