In the ever-evolving landscape of chemical education, the comprehension of advanced concepts like the Master N3 Lewis structure stands as a pillar for aspiring chemists. The complexity of determining the electron distribution in complex molecules can often overwhelm beginners. However, this task becomes significantly more approachable with a clear understanding of molecular geometry and electron pair arrangements. This article delves into the intricate yet fascinating details of the Master N3 Lewis structure, providing both novices and seasoned professionals with a comprehensive guide.
Key Insights
- Understanding the Master N3 Lewis structure is pivotal for grasping molecular geometry.
- The application of the octet rule and formal charge calculations is fundamental in this process.
- Proper depiction of lone pairs and bonding electrons leads to accurate molecular configurations.
Foundations of Lewis Structure
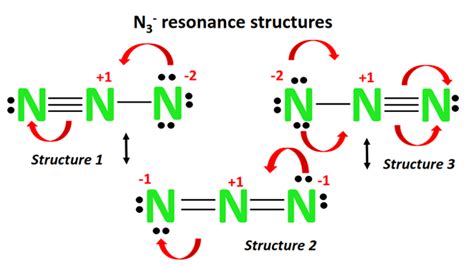
At its core, the Master N3 Lewis structure is a depiction of how nitrogen atoms share electrons to form a stable molecule. The first step involves counting the total valence electrons, which for nitrogen (N) is five. When three nitrogen atoms interact, the challenge lies in forming three-way bonds while adhering to the octet rule, which states that atoms seek eight valence electrons to achieve stability.This foundational understanding necessitates an analytical approach, often leading to trial and error in structure formations. Initially, one might consider three nitrogen atoms forming a linear structure, but this does not adequately distribute electrons to satisfy the octet rule for every atom. Hence, the Lewis structure necessitates an unconventional depiction compared to simpler molecules.
Practical Application of Formal Charge
A critical element in constructing an accurate Master N3 Lewis structure is calculating the formal charge on each atom. The formal charge is determined using the formula: Formal Charge = Valence Electrons - (Non-bonding Electrons + 1⁄2 Bonding Electrons). For nitrogen, this means calculating based on the electrons involved in each potential bond configuration.An effective strategy in this context is to distribute electrons to achieve a formal charge of zero where possible, indicating a stable molecule. In the Master N3 structure, achieving this necessitates forming a trigonal planar arrangement with each nitrogen atom forming three bonds. Consequently, one nitrogen atom must also accommodate a lone pair to balance the electron distribution. This advanced method highlights the criticality of formal charge calculations in achieving precise molecular representations.
What challenges arise when constructing the Master N3 Lewis structure?
The primary challenge lies in correctly distributing electrons to satisfy the octet rule for all nitrogen atoms while forming three-way bonds. This requires careful consideration of bond configurations and lone pair placements.
How important is the concept of formal charge in the Lewis structure construction?
The concept of formal charge is critical. It helps in ensuring that the molecule’s electron distribution achieves stability, leading to an accurate and stable molecular configuration.
Through a nuanced understanding of the Master N3 Lewis structure, chemists can unlock deeper insights into complex molecular formations, aiding in the design of advanced chemical compounds and fostering innovations in fields ranging from pharmaceuticals to materials science. This exploration not only underscores the importance of molecular geometry but also emphasizes the practical application of theoretical concepts in chemical education and research.