Understanding the ground state electron configuration of elements is foundational for anyone delving into chemistry, physics, or related fields. This guide aims to demystify the concept, providing you with step-by-step guidance, actionable advice, and practical solutions to ensure you grasp these secrets efficiently.
Understanding Electron Configurations
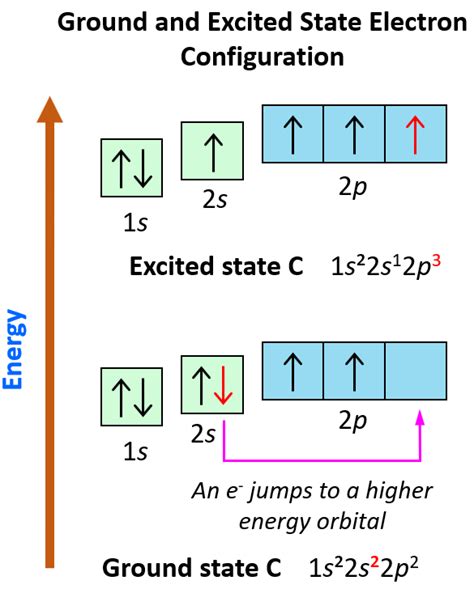
Electron configurations represent the distribution of electrons within an atom's orbitals. The ground state electron configuration of an element tells us how electrons are arranged in the orbitals when the atom is in its lowest energy state. This is crucial for predicting chemical behavior, atomic stability, and various other chemical properties.
Struggling to understand this might leave you puzzled and overwhelmed. Fear not! This guide is here to clarify the complex world of electron configurations, breaking down the core concepts into digestible pieces. Let's dive into a problem-solution approach to help you grasp these secrets effectively.
Let’s start by addressing some common challenges users face when learning about ground state electron configurations:
One of the biggest hurdles is understanding the rules governing electron placement. Another is determining the correct electron configurations without using complex memorization techniques. By providing practical examples and a structured approach, this guide aims to simplify these tasks and make them intuitive.
Quick Reference
Quick Reference
- Immediate action item: Memorize the Aufbau principle: electrons fill the lowest energy orbitals first.
- Essential tip: Use the periodic table to determine the electron configuration of elements by following the order of increasing atomic number.
- Common mistake to avoid: Confusing the notation for electron configurations (e.g., 2p6 vs. 2p2) and overlooking the Hund’s rule.
Detailed How-To: Aufbau Principle
The Aufbau principle is one of the foundational rules in understanding electron configurations. This principle states that electrons occupy the lowest energy orbital available before filling higher ones. This rule is crucial because it adheres to the natural tendency of electrons to arrange themselves in the most stable configuration, thereby minimizing the atom's energy.
Here’s how to apply it:
- Start with hydrogen: Hydrogen has one electron which goes into the 1s orbital.
- Helium to neon: The next six electrons fill the 1s and 2s orbitals fully, followed by the 2p orbitals which accommodate the remaining two electrons up to a total of eight electrons. This is why the electron configuration for neon is 1s2 2s2 2p6.
- Beyond neon: Start with the next orbital sequence. For instance, sodium begins filling the 3s orbital after the neon configuration.
To summarize:
Hydrogen: 1s1
Helium: 1s2
Lithium: 1s2 2s1
Beryllium: 1s2 2s2
Boron: 1s2 2s2 2p1
Carbon: 1s2 2s2 2p2
Nitrogen: 1s2 2s2 2p3
Oxygen: 1s2 2s2 2p4
Fluorine: 1s2 2s2 2p5
Neon: 1s2 2s2 2p6
Detailed How-To: Hund’s Rule
Another key principle is Hund’s rule. This rule addresses the electron placement in orbitals of equal energy, such as the 2p orbitals. According to Hund’s rule, electrons will fill these orbitals singly first before any pairing occurs, and all electrons in singly occupied orbitals have the same spin direction.
This helps in achieving a more stable and lower energy state for the atom. Here’s how to apply it:
- Identify orbitals of equal energy: For example, in the 2p orbitals.
- Place electrons singly first: Place each electron in a different 2p orbital while maintaining parallel spins.
- Only pair up when all orbitals are singly occupied: Once all 2p orbitals are singly occupied, then electrons will start to pair up.
So for oxygen, the electron configuration would be 1s2 2s2 2p4. Following Hund’s rule ensures maximum spin and stability.
Practical FAQ
What happens if I don’t follow the Aufbau principle?
If you don’t follow the Aufbau principle, your electron configuration won’t reflect the actual ground state of the atom. This could lead to incorrect predictions about the atom’s chemical properties and reactivity.
For example, if you mistakenly fill the 3p orbital before the 3s orbital, the atom will not be in its lowest energy state. Such errors can lead to incorrect conclusions about bonding behaviors and interactions with other atoms.
Why do electrons in s orbitals fill first?
Electrons fill s orbitals before p, d, and f orbitals because they have lower energy levels. Lower energy orbitals are more stable, and electrons prefer to occupy these positions to minimize the atom’s overall energy. This is a reflection of nature’s tendency to achieve the lowest possible energy state.
For example, the 1s orbital is filled before the 2s and 2p orbitals, as the 1s orbital has less energy, leading to a more stable atom.
How do I know which electron configuration to use when elements have exceptions?
Most elements follow the standard electron configurations based on the Aufbau principle and Hund’s rule. However, some elements, particularly transition metals and inner transition metals, have exceptions. These exceptions often involve half-filled or fully-filled d orbitals providing extra stability.
For instance, Chromium (Cr) has an electron configuration of [Ar] 4s1 3d5 instead of the expected [Ar] 4s2 3d4 because the 3d5 configuration is more stable due to half-filling.
These exceptions are crucial to remember, particularly in advanced chemistry courses where these details matter for understanding chemical behaviors and bonding.
To wrap up, mastering ground state electron configurations begins with understanding and applying these key principles. The key is to focus on the rules: Aufbau, Hund’s, and Pauli Exclusion, along with periodic trends. With practice and these foundational steps, you’ll gain confidence and clarity in tackling electron configurations.
Remember, practical application and revisiting these concepts through exercises will reinforce your learning. Happy studying, and may the secrets of ground state electron configurations become clear to you!