Understanding the CHCL3 Lewis Structure: A Deep Dive into Chloroform’s Molecular Blueprint
The CHCL3 molecule, commonly known as chloroform, holds a pivotal place in chemistry due to its historical significance and practical applications. Grasping its CHCL3 Lewis structure is crucial for comprehending its bonding and molecular geometry. Here, we delve into chloroform’s molecular blueprint, providing a clear and expert perspective on its fundamental structure.
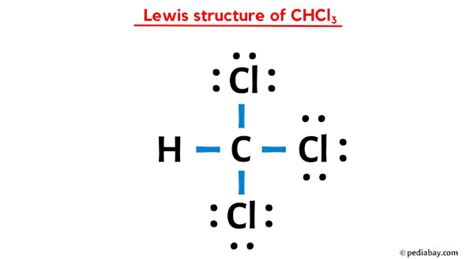
To begin, let’s understand the molecular structure of CHCL3. This molecule consists of one carbon atom bonded to three chlorine atoms and one hydrogen atom. The Lewis structure, a representation that depicts the arrangement of atoms and valence electrons, helps us visualize how these atoms form bonds and share electrons. To construct the CHCL3 Lewis structure, we must first count the valence electrons available to us. Carbon contributes 4, each chlorine contributes 7, and hydrogen contributes 1, summing up to a total of 26 valence electrons for the entire molecule.
Key Insights
- Primary insight with practical relevance: The CHCL3 Lewis structure reveals the tetrahedral geometry, crucial for understanding the molecule's reactivity and interactions.
- Technical consideration with clear application: The molecule’s central carbon atom forms four sp3 hybridized orbitals, allowing for stable tetrahedral molecular geometry.
- Actionable recommendation: To further understand the molecule’s behavior, consider its resonance structures and formal charges.
Construction of the CHCL3 Lewis Structure
Constructing the CHCL3 Lewis structure involves several steps. Start by placing the least electronegative atom at the center, which in this case is carbon. Surround carbon with the three chlorine atoms and one hydrogen atom. Begin by forming single bonds between carbon and each chlorine, using up 24 valence electrons. Place the remaining 2 electrons on the carbon atom to satisfy the octet rule for carbon. Since chlorine is more electronegative, it will not accept additional electrons beyond the bond pairs, ensuring a stable structure.
Geometrical Configuration
The CHCL3 molecule adopts a tetrahedral geometry due to the four sp3 hybridized orbitals surrounding the central carbon atom. This arrangement minimizes electron-pair repulsion and maintains stability. Each chlorine atom occupies a vertex of the tetrahedron, with the hydrogen atom situated at the remaining vertex. This geometrical arrangement allows chloroform to exhibit specific chemical properties, such as its relatively high boiling point and its polarity, which affect its solubility and reactivity in various chemical environments.
Why is understanding the CHCL3 Lewis structure important?
Understanding the CHCL3 Lewis structure is important because it provides insight into the molecule's bonding characteristics, molecular geometry, and reactivity, which are essential for studying its chemical properties and applications in various fields.
How does the geometry of CHCL3 influence its chemical behavior?
The tetrahedral geometry of the CHCL3 molecule affects its chemical behavior by determining the spatial arrangement of the atoms and the molecule’s dipole moment. This, in turn, influences chloroform’s interactions with other molecules, including its solubility in polar solvents and its reactivity in chemical reactions.
In conclusion, the CHCL3 Lewis structure reveals the molecular blueprint that underpins chloroform’s unique chemical properties. By constructing and analyzing its Lewis structure, we gain a comprehensive understanding of its bonding, geometry, and reactivity, paving the way for further exploration and application in various chemical domains.